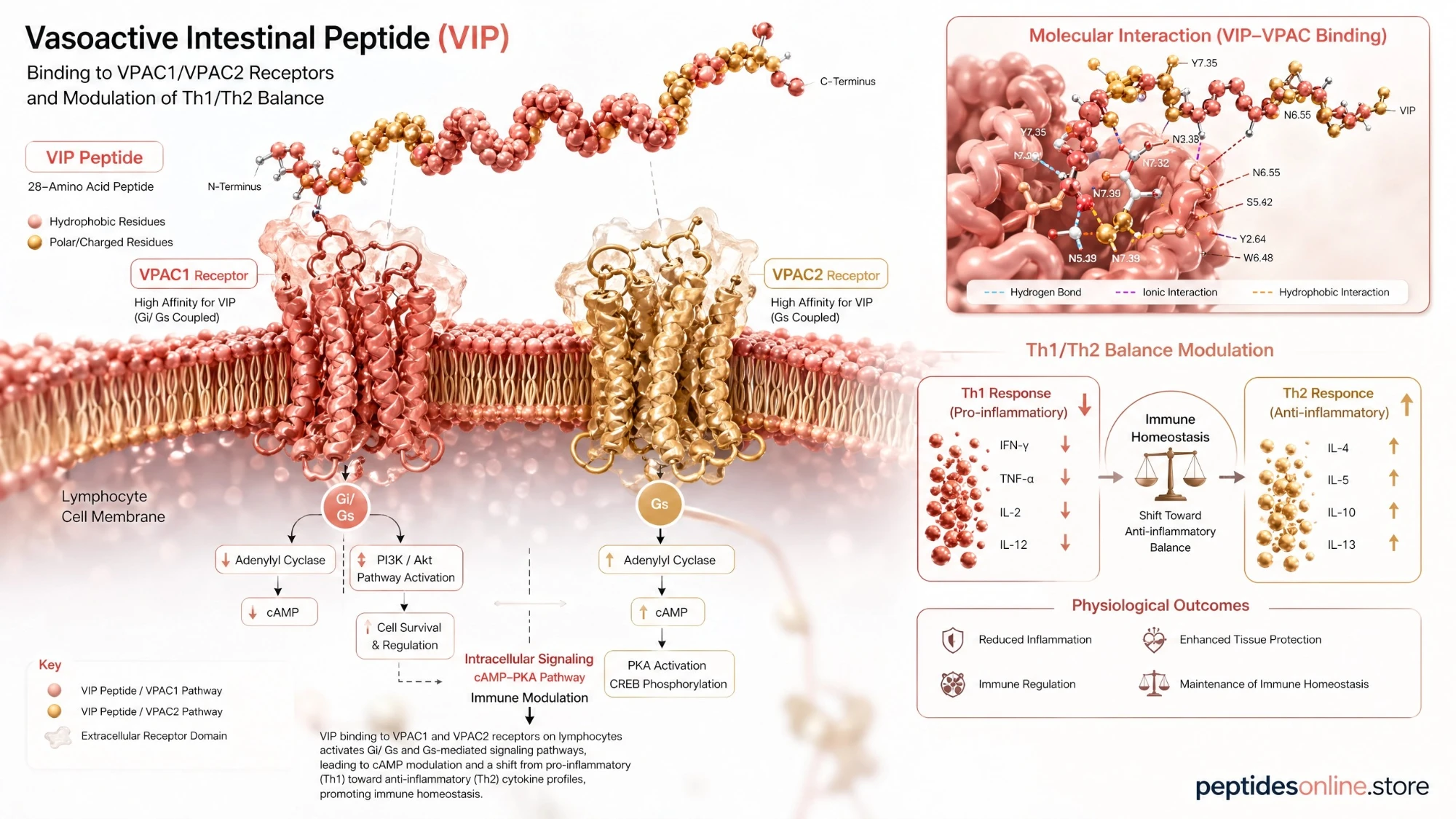
VIP (Vasoactive Intestinal Peptide)
Our VIP is produced in an ISO 9001:2015 certified facility using advanced Solid-Phase Peptide Synthesis (SPPS) technology. We ensure an ultra-high chemical purity of ≥98%, rigorously validated through High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS). This stringent quality control process guarantees a product free from trifluoroacetic acid (TFA) residues and truncated peptide sequences, ensuring the molecular integrity and reproducible biological activity essential for high-fidelity immunological research and therapeutic development assays.
Research Applications & Mechanisms
Neuro-Immune Axis Modulation
Vasodynamic Stability & Microcirculation
Cellular Cytoprotection & Homeostasis
G-Protein Coupled Receptor (GPCR) Signaling
Technical Specifications
| Parameter | Laboratory Standard |
| Manufacturing Standard | ISO 9001:2015 Certified Facility |
| Production Quality | GMP-Level Compliance |
| Purity (HPLC) | ≥99% (Pharmaceutical Grade) |
| Physical Appearance | Fine, Smooth & Flat Lyophilized Cake |
| Molecular Weight | 3325.8 Da |
| Solubility | Soluble in Water or 1% Acetic Acid |
| Batch Control | MS & HPLC Analysis Verified |
| Filtration | 0.22 μm sterile filtered |
Key Features & Quality Assurance
-
Pharmaceutical-Grade Lyophilization: Our proprietary vacuum-sealing process results in a perfectly smooth, flat, and delicate lyophilized cake. This professional structure ensures rapid reconstitution and maximum molecular stability.
-
ISO 9001 Quality Management: Every production cycle is governed by strict international standards to guarantee batch-to-batch consistency for reproducible research.
-
GMP Compliance: Manufactured in a certified sterile environment to eliminate cross-contamination and ensure the highest chemical identity.
Reconstitution Guide (Important)
To preserve the peptide’s biological activity, follow these protocols:
-
Preparation: Allow the vial to reach room temperature. Centrifuge at 30-60 seconds to ensure the cake is positioned at the bottom.
-
Solvent Selection: Reconstitute with Sterile Water or Bacteriostatic Water. For long-term stock solutions, a weak acid (e.g., 1% acetic acid) may improve solubility.
-
Technique: Gently roll the diluent down the side of the vial. DO NOT SHAKE; swirl gently until the solution is clear.
Storage & Handling
-
Long-term: Store at -20°C (Stable for up to 24 months).
-
Short-term: Stable at 2–8°C for up to 3 months.
-
Post-Reconstitution: Store reconstituted solution at 2–8°C and utilize within 10-14 days.
Shipping & Documentation
-
Global Logistics: Discreet, temperature-controlled packaging.
-
Verified Quality: Batch-specific COA (Certificate of Analysis) and HPLC/MS reports are provided with every order.
- Reshipment Policy: We provide a guaranteed reshipment policy for any lost or customs-intercepted orders (terms apply).
⚠️ Research Disclaimer
For Laboratory Research and Analytical Purposes Only. VIP is NOT a drug, food, or cosmetic and is strictly not intended for human or veterinary consumption.
Scientific Resources & References
-
National Center for Biotechnology Information (NCBI): Vasoactive Intestinal Peptide Summary (CID 16132284)
-
ScienceDirect (Elsevier): Research Topics on Vasoactive Intestinal Peptide Functions
🔍 Deep Dive Research
Expand your laboratory protocols with our technical insights:
-
[Technical Guide]: How to Reconstitute and Store Lyophilized Peptides for Maximum Potency
-
[Neuroscience Study]: The Role of VIP in Circadian Rhythm and Neuro-Immunology
-
[Quality Standards]: The Importance of GMP-Grade Manufacturing in Peptide Synthesis





Jermarwag –
Everything arrived fine.