Retatrutide (LY3437943)
Retatrutide (internally designated as LY3437943) represents the newest frontier in multi-receptor pharmacology. It is a synthetic 39-amino acid peptide that functions as a powerful triple agonist, simultaneously targeting the Glucagon-Like Peptide-1 (GLP-1) receptor, the Glucose-Dependent Insulinotropic Polypeptide (GIP) receptor, and the Glucagon receptor (GCGR). In advanced metabolic and obesity research, Retatrutide is uniquely distinguished by its integration of the glucagon receptor pathway. By activating GCGR, it stimulates energy expenditure and promotes hepatic lipid metabolism, which, when combined with the glycemic control of GLP-1 and GIP, providing researchers with a non-linear model for investigating weight loss and metabolic remodeling.
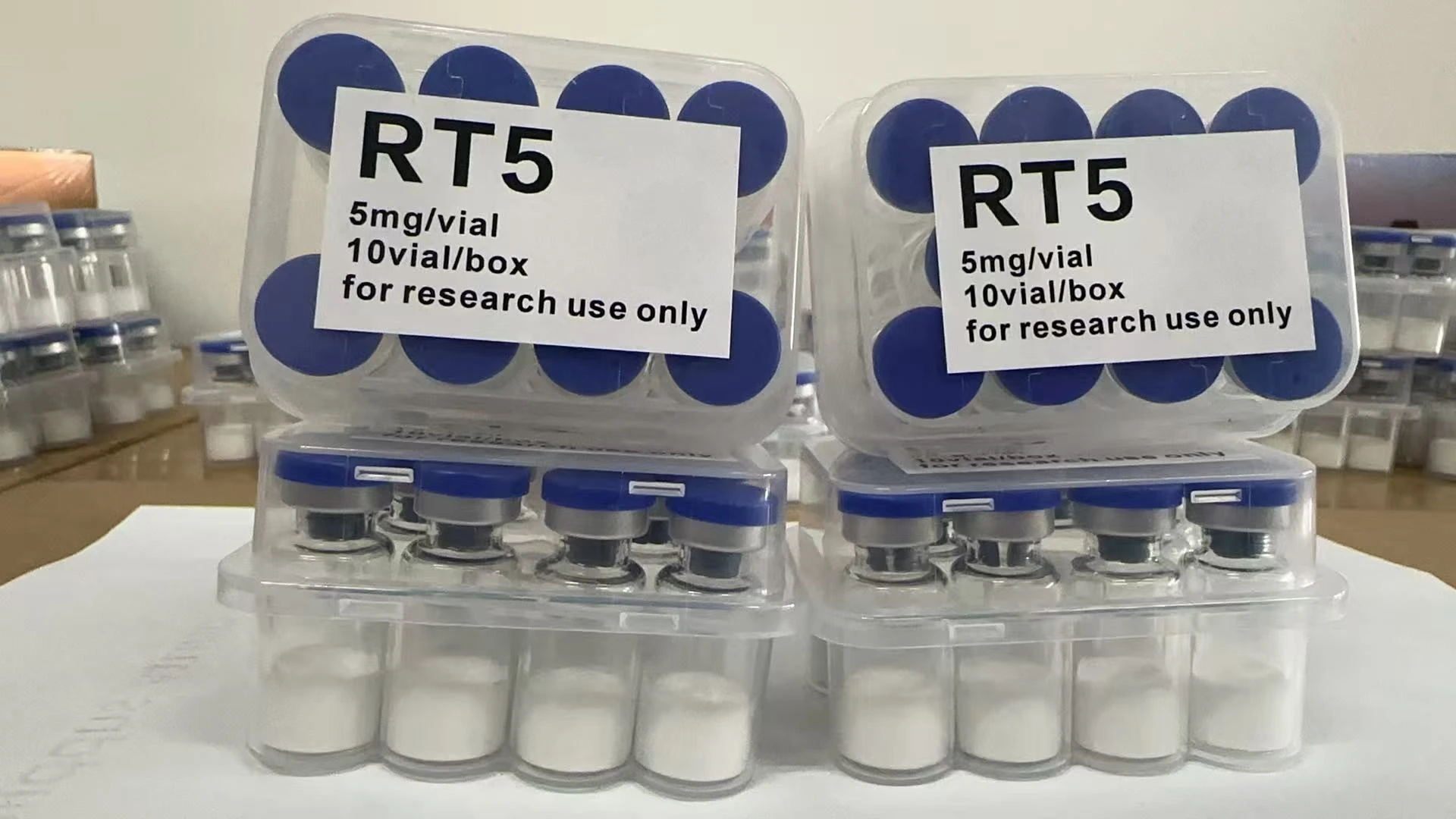
Our Retatrutide is synthesized in an ISO 9001:2015 certified and GMP-grade manufacturing facility. Utilizing state-of-the-art solid-phase synthesis and high-resolution multi-stage HPLC purification, we achieve an ultra-high chemical purity of ≥99%. This meticulous production standard ensures the peptide is free from truncated sequences and synthesis by-products, guaranteeing the consistency and stability required for high-stakes studies into triple-receptor synergy and cellular bioenergetics.

Research Applications & Mechanisms
Triple-Receptor Synergy (GLP-1/GIP/GCGR)
Investigating the potentiation of metabolic signaling when the glucagon receptor is activated alongside incretin receptors to increase basal metabolic rate.
Thermogenesis & Energy Expenditure
Researching the molecular pathways through which Retatrutide stimulates thermogenesis and fat oxidation in experimental metabolic models.
Hepatic Fat Mobilization
Studying the influence of GCGR activation on liver lipid homeostasis and its potential in metabolic dysfunction-associated steatohepatitis (MASH) research.
Advanced Glycemic Homeostasis
Exploring the complex interplay between triple-agonism and pancreatic beta-cell stability for superior glucose regulation in research models.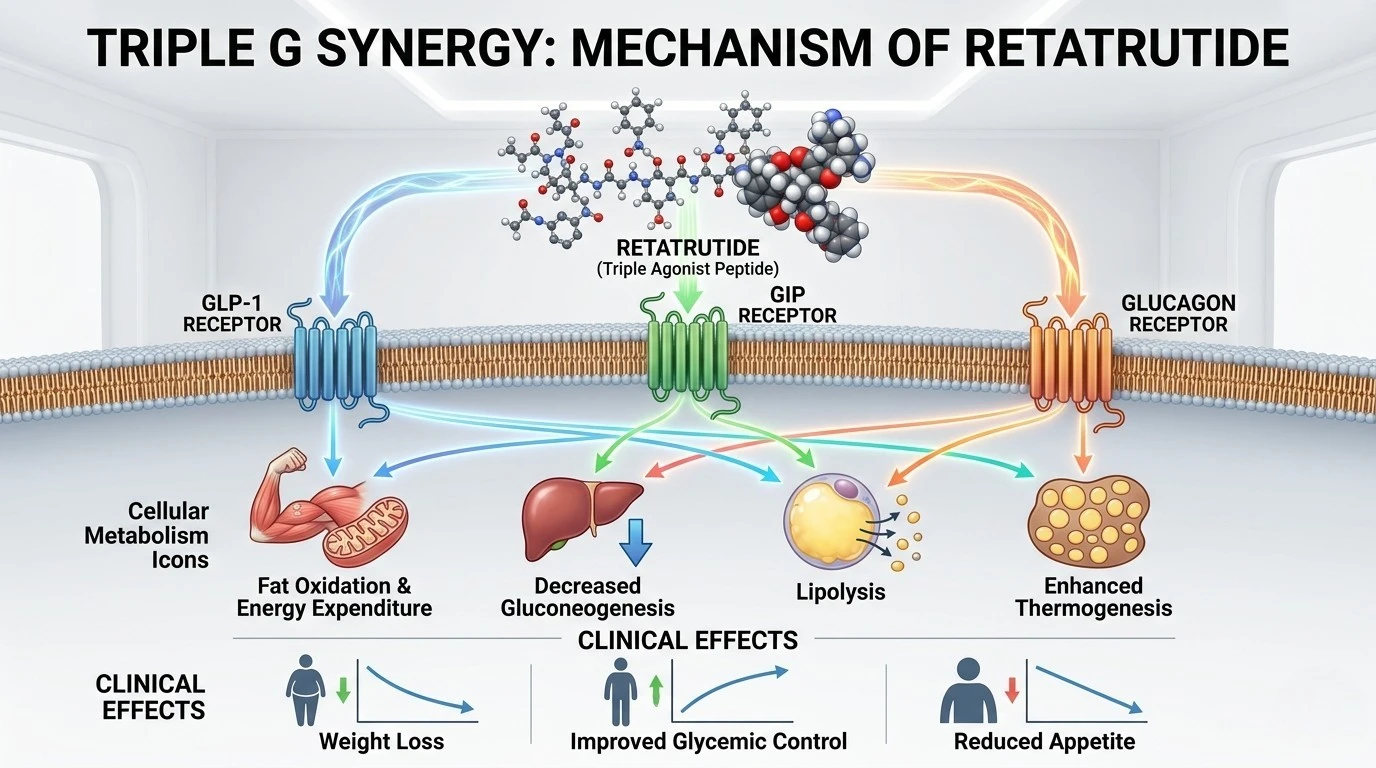
Technical Specifications
| Parameter | Laboratory Standard |
| Compound | Retatrutide (LY3437943) |
| Agonist Type | Triple GLP-1R / GIPR / GCGR Agonist |
| Production Quality | GMP-Level Compliance |
| Purity (HPLC) | ≥99% (Pharmaceutical Grade) |
| Physical Appearance | Fine, Smooth & Flat Lyophilized Cake |
| Sequence | 39-Amino Acid Stabilized Fragment |
| Concentration | 5mg / 10mg / 15mg / 20mg / 30mg / 40mg / 50mg / 60mg / 100mg |
| Solubility | Highly Soluble in Water / Bacteriostatic Water |
| Batch Control | MS & HPLC Analysis Verified |
Key Features & Quality Assurance
Pharmaceutical-Grade Lyophilization
Our proprietary vacuum-sealing process ensures the Retatrutide powder surface is perfectly smooth, flat, and delicate. This uniform “cake” structure optimizes reconstitution and ensures the structural integrity of the complex triple-agonist sequence.
Structural Fidelity & Stability
Every batch undergoes strict Mass Spectrometry (MS) to verify the sequence and HPLC to confirm purity exceeds research requirements, ensuring maximal receptor binding affinity in experimental environments.
Reconstitution & Storage
Reconstitution Guide
-
Preparation: Allow the vial to reach room temperature. Centrifuge for 30-60 seconds to ensure the Retatrutide cake is at the base.
-
Solvent: Reconstitute with Sterile Water or Bacteriostatic Water.
-
Technique: Gently introduce the diluent against the vial wall. DO NOT SHAKE; swirl gently until the solution is clear and transparent.
Storage Protocols
-
Long-term: Store at -20°C (Maintains potency for up to 24 months).
-
Short-term: Stable at 2–8°C for up to 3 months.
-
Post-Reconstitution: Store at 2–8°C and utilize within 14-21 days for optimal results.
Shipping & Documentation
-
Global Logistics: Shipped in discreet, temperature-controlled packaging to protect the Retatrutide molecular integrity.
-
Verified Quality: A batch-specific COA and HPLC/MS reports are available for every order.
-
Reshipment Policy: We provide a guaranteed reshipment policy for any lost or customs-intercepted orders (terms apply).
⚠️ Research Disclaimer
Product-Specific Safety Notice: This Retatrutide (LY3437943) is strictly for laboratory research and analytical purposes. It is NOT intended for human or veterinary administration. All research must be conducted by qualified professionals.
Scientific Resources & References
-
National Center for Biotechnology Information (NCBI): Compound Summary – Retatrutide (LY3437943; GLP-1/GIP/Glucagon triple receptor agonist)
-
PubMed – NIH: Clinical studies on Retatrutide (LY3437943) in obesity and metabolic disease
🔍 Deep Dive Research
-
[Technical Guide]: How to Reconstitute and Store Lyophilized Peptides for Maximum Potency
-
[Metabolic Study]: The Triple Agonist Frontier: Retatrutide and the Future of Energy Expenditure Research






dsl –
Delivered safely.