The landscape of metabolic research is witnessing a seismic shift. While the scientific community is still integrating the success of Semaglutide (GLP-1) and Tirzepatide (GLP-1/GIP), a new molecule—Retatrutide (LY3437943)—is setting a new pharmacological benchmark. As research transitions from dual-agonism to Triple-Agonism, understanding the multi-receptor synergy of Retatrutide is essential for any advanced laboratory study.
1. The “Triple G” Mechanism: A New Era in Pharmacology
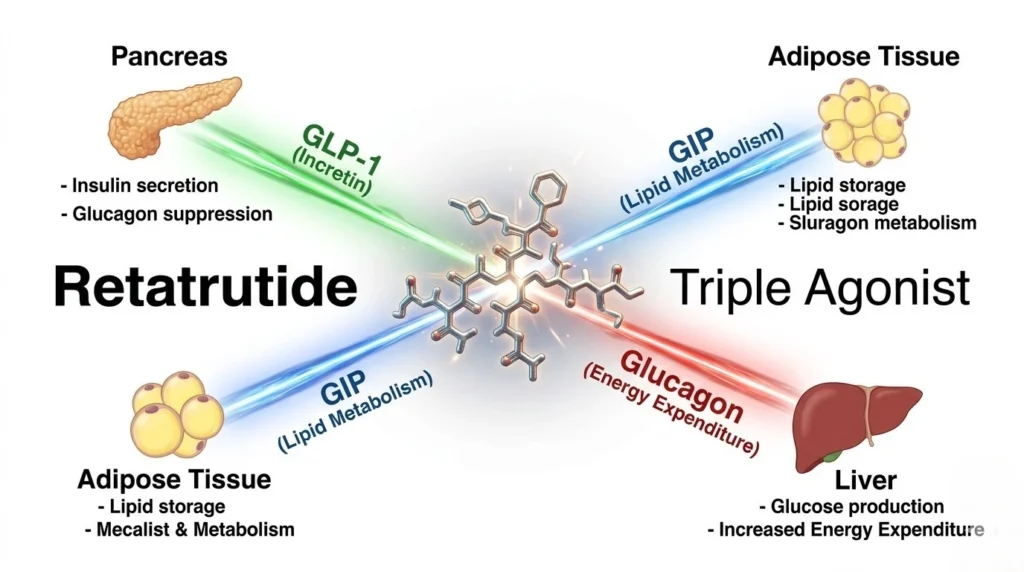
Retatrutide is a 39-amino acid synthetic peptide that functions as a unimolecular triple agonist. It is often referred to in industry circles as “Triple G” because it simultaneously targets three distinct nutrient-stimulated hormone receptors. Each pathway offers a unique contribution to metabolic homeostasis:
A. GLP-1 (Glucagon-Like Peptide-1)
Primarily secreted in the gut, GLP-1’s role is well-documented: it enhances glucose-dependent insulin secretion, slows gastric emptying, and induces satiety via the central nervous system. In Retatrutide, the GLP-1 component provides the foundational “anorectic” effect.
B. GIP (Glucose-Dependent Insulinotropic Polypeptide)
GIP complements GLP-1 by further improving insulin sensitivity and, crucially, modulating lipid metabolism. Research suggests that GIP agonism may help buffer the potential nausea associated with high-dose GLP-1, allowing for better tolerability in escalated research protocols.
C. Glucagon Receptor (GCGR) — The Game Changer
The inclusion of Glucagon agonism is what elevates Retatrutide above its predecessors. Historically, glucagon was viewed as a mere antagonist to insulin. Modern peptide science now reveals that controlled GCGR activation increases energy expenditure (thermogenesis) and directly promotes lipolysis in the liver. By adding this third pillar, Retatrutide doesn’t just manage caloric intake; it actively modulates the metabolic rate.
2. Comparative Analysis: Retatrutide vs. Tirzepatide
To understand the value of Retatrutide in a research setting, a direct comparison with the current gold standard, Tirzepatide, is necessary.
| Feature | Semaglutide | Tirzepatide | Retatrutide |
| Receptor Targets | GLP-1R | GLP-1R / GIPR | GLP-1R / GIPR / GCGR |
| Molecule Type | Mono-agonist | Dual-agonist | Triple-agonist |
| Metabolic Impact | Appetite suppression | Satiety + Insulin Sensitivity | Satiety + Sensitivity + Thermogenesis |
| Liver Fat Reduction | Moderate | High | Significant (Up to 80-85% reduction) |
| Energy Expenditure | Neutral | Neutral | Elevated via GCGR pathway |
In recent Phase II clinical research, Retatrutide demonstrated weight reduction efficacy that surpassed the 12-month benchmarks of both Semaglutide and Tirzepatide within a shorter timeframe. For researchers, this suggests a more potent synergistic threshold when all three metabolic pathways are activated simultaneously.
3. Implications for NAFLD and Hepatic Research
One of the most promising frontiers for Retatrutide is Hepatology. Chronic metabolic studies often struggle with the complexities of liver fat accumulation, formerly known as NAFLD (Non-Alcoholic Fatty Liver Disease), now categorized as MASLD.
Retatrutide’s ability to “scavenge” hepatic lipids is a direct result of glucagon receptor activation in the liver. By stimulating these receptors, Retatrutide promotes the oxidation of fatty acids within the mitochondria. Preliminary data indicates that Retatrutide can reduce liver fat content by over 80% in specific research models, potentially offering a pathway to reverse steatosis more effectively than any existing dual-agonist.
4. Laboratory Considerations: Reconstitution and Stability
For investigators handling Retatrutide, precision is paramount. As a triple-agonist, the structural integrity of its 39-amino acid chain is highly dependent on its environment.
- Solubility: Retatrutide is generally lyophilized for stability. It should be reconstituted using Bacteriostatic Water (0.9% Benzyl Alcohol) to ensure sterility during multi-dose research.
- pH Sensitivity: The triple-binding motifs are sensitive to pH fluctuations. Maintaining a near-neutral pH during reconstitution is vital to prevent peptide aggregation.
- Storage: Post-reconstitution, the peptide must be stored at 2°C – 8°C (36°F – 46°F). Avoid agitation or vigorous shaking, which can denature the delicate peptide bonds.
Research Opportunity: For advanced comparative studies, our Retatrutide Analytical Standards are synthesized to ≥ 99% purity, verified via HPLC and Mass Spectrometry.
5. Conclusion
Retatrutide represents the next logical step in the evolution of metabolic pharmacology. By bridging the gap between calorie restriction and active energy expenditure, it provides a comprehensive tool for studying the complexities of human metabolism. As the peptide industry moves toward more personalized and multi-targeted interventions, Retatrutide stands as the current frontier of what is possible.
Technical Profile Summary
- CAS Number: 2381089-83-2
- Molecular Weight: 4731.33 g/mol
- Sequence: 39-amino acid backbone with fatty acid acylation for extended half-life.
- Purity Standards: ≥ 99% via HPLC/MS.
- Primary Research Use: Metabolic syndrome, hepatic lipid signaling, and obesity research models.
Peer-Reviewed Scientific References
The New England Journal of Medicine (NEJM): Triple–Hormone–Receptor Agonist Retatrutide for Obesity
The Lancet: Retatrutide for the treatment of type 2 diabetes
Nature Medicine: Evolution of incretin-based therapies in metabolic disease
ScienceDirect: The role of Glucagon agonism in energy expenditure
NOTE: These products are intended for laboratory research use only. This peptide is not intended for personal use. Please review and adhere to our Terms and Conditions before ordering.