BACTERIOSTATIC WATER (Bac-water)
Bacteriostatic Water (Bac-water) is a high-purity, sterile-filtered water preparation containing 0.9% Benzyl Alcohol ($C_7H_8O$) as a bacteriostatic preservative. In the landscape of advanced peptide and protein research, Bac-water is the gold-standard solvent for the reconstitution of lyophilized compounds. Unlike standard sterile water, which must be discarded after a single use due to the risk of bacterial contamination, the presence of benzyl alcohol in Bac-water effectively inhibits the growth of most potential contaminating bacteria. This allows researchers to utilize a single vial for multiple experimental draws over an extended period. It provides a stable, pH-balanced environment to investigate the metabolic flux and long-term bio-activity of synthetic peptides while maintaining the highest standards of laboratory hygiene and data integrity.
Our Bac-water is produced in an ISO 9001:2015 certified environment utilizing USP-grade components and Ultra-Pure Type I water. The solution undergoes rigorous 0.22μm membrane filtration and vacuum sealing to ensure total sterility. This ensures the solvent is free from pyrogens, heavy metals, and organic impurities, guaranteeing that the chemical structure of sensitive research peptides remains unaltered during the reconstitution process.
Research Applications & Mechanisms
Stabilization of Multi-Dose Research
Investigating the long-term integrity of reconstituted peptides by preventing bacterial degradation in multi-draw experimental designs.
Preservation of Peptide Potency
Researching the influence of bacteriostatic environments on the shelf-life of dissolved amino acid chains compared to non-preserved aqueous solutions.
Standardized Solvent for In-Vitro Assays
Utilizing a consistent, preserved solvent to minimize experimental variables caused by microbial contamination in sensitive biochemical pathways.
Optimization of Laboratory Resource Management
Studying the cost-efficiency and procedural safety of using multi-use bacteriostatic solvents in high-throughput peptide screening.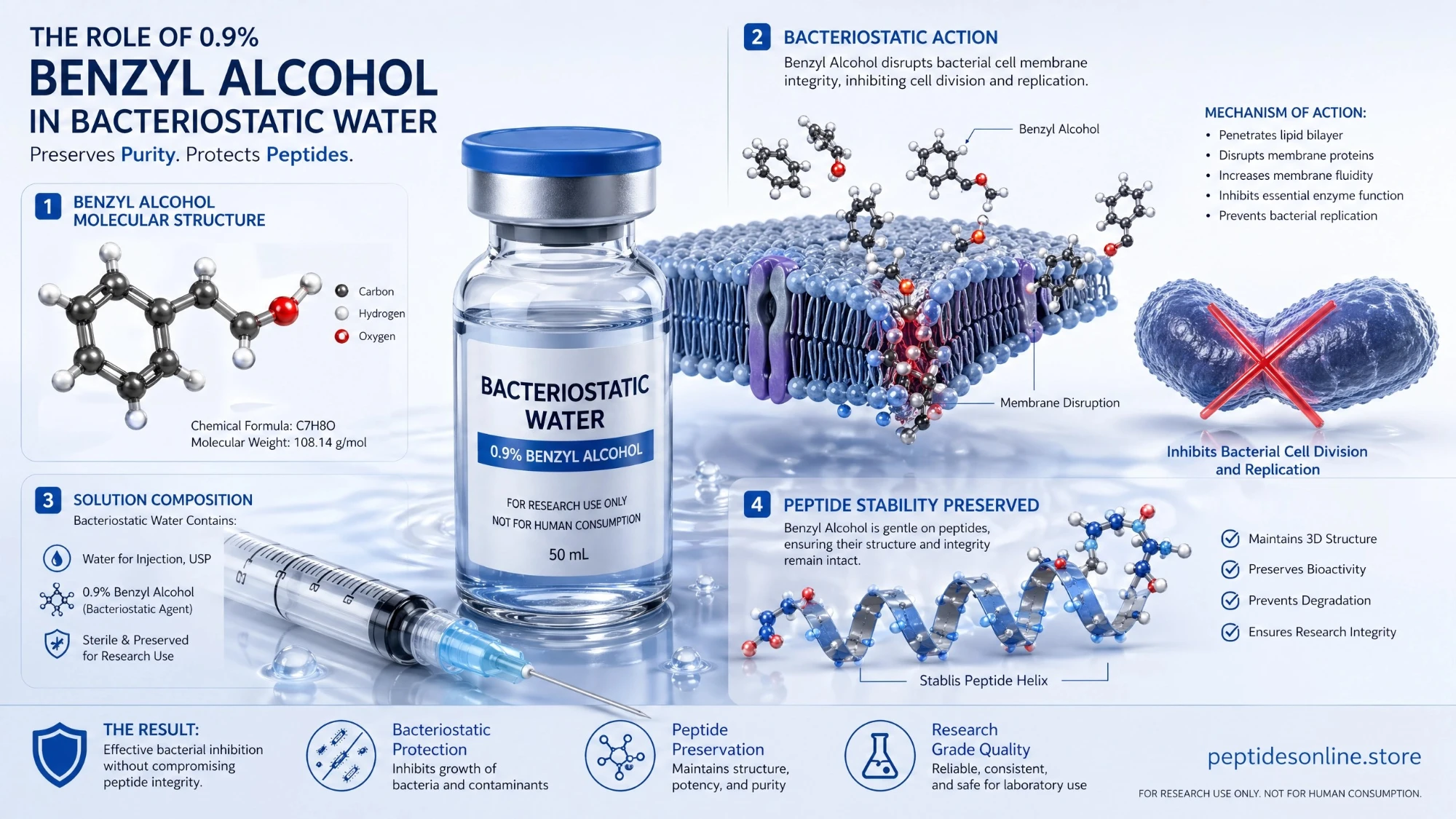
Technical Specifications
| Parameter | Laboratory Standard |
| Product | Bacteriostatic Water (BA / WAC) |
| Preservative | 0.9% Benzyl Alcohol |
| Base | USP-Grade Sterile Water for Injection |
| Physical Appearance | Clear, Colorless Liquid |
| pH Range | 5.7 (4.5 to 7.0) |
| Concentrations | Available in 3ml / 5ml / 10ml vials |
| Sterility | Validated 0.22μm Membrane Filtration |
| Container | Medical-Grade Borosilicate Glass |
Key Features & Quality Assurance
Certified Multi-Use Integrity
Our Bac-water is formulated to maintain its bacteriostatic properties for up to 28 days after the initial vial entry, provided proper aseptic techniques are maintained.
Pyrogen-Free & Deionized
Every batch is tested to ensure zero endotoxin levels and ultra-low conductivity, preventing any unintended ionic interference with complex peptide-receptor binding studies.
Usage & Storage Guidelines
Reconstitution Best Practices
-
Aseptic Technique: Always wipe the rubber stopper with a 70% isopropyl alcohol swab before every needle entry.
-
Volume Precision: Utilize a sterile syringe to draw the exact volume required for your peptide concentration (e.g., 1ml of Bac-water for a 5mg peptide vial creates a 5mg/ml concentration).
-
Solubility: Introduce the Bac-water slowly against the vial wall to prevent protein foaming or shearing.
Storage Protocols
-
Unopened: Store at room temperature (15–25°C) away from direct sunlight.
-
Opened: Once the seal is broken, it is recommended to store at 2–8°C to further enhance stability.
-
Shelf Life: Maintains laboratory-grade potency for up to 24 months (unopened).
Shipping & Documentation
-
Global Logistics: Shipped in break-resistant, discreet packaging. Bac-water is stable at ambient temperatures during global transit.
-
Verified Quality: A lot-specific Certificate of Analysis (COA) is provided with every shipment.
-
Reshipment Policy: We provide a guaranteed reshipment policy for any lost or customs-intercepted orders (terms apply).
⚠️ Research Disclaimer
Product-Specific Safety Notice: This Bacteriostatic Water is strictly for laboratory research and analytical purposes. It is NOT intended for human or veterinary administration. All research must be conducted by qualified professionals.
Scientific Resources & References
-
Pubmed: Critical Aspects of pH Measurement for Bacteriostatic Water for Injection.
-
Journal of Pharmaceutical Sciences: Effects of Benzyl Alcohol on Aggregation of Recombinant Human Interleukin-1-Receptor Antagonist in Reconstituted Lyophilized Formulations
🔍 Deep Dive Research
-
[Technical Guide]: Sterile Water vs. Bacteriostatic Water: Choosing the Right Solvent for Your Research
-
[Stability Study]: The 28-Day Rule: Maintaining Sterility in Multi-Use Research Vials




qaysar –
No issues with the order.