In the unregulated landscape of research chemicals, “99% Purity” is a claim easily made but rarely verified by the buyer. For a serious investigator, the only acceptable proof of quality is a Certificate of Analysis (COA) backed by two pillars of analytical chemistry: High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS).
Understanding how to read these reports is the final line of defense against substandard research materials.

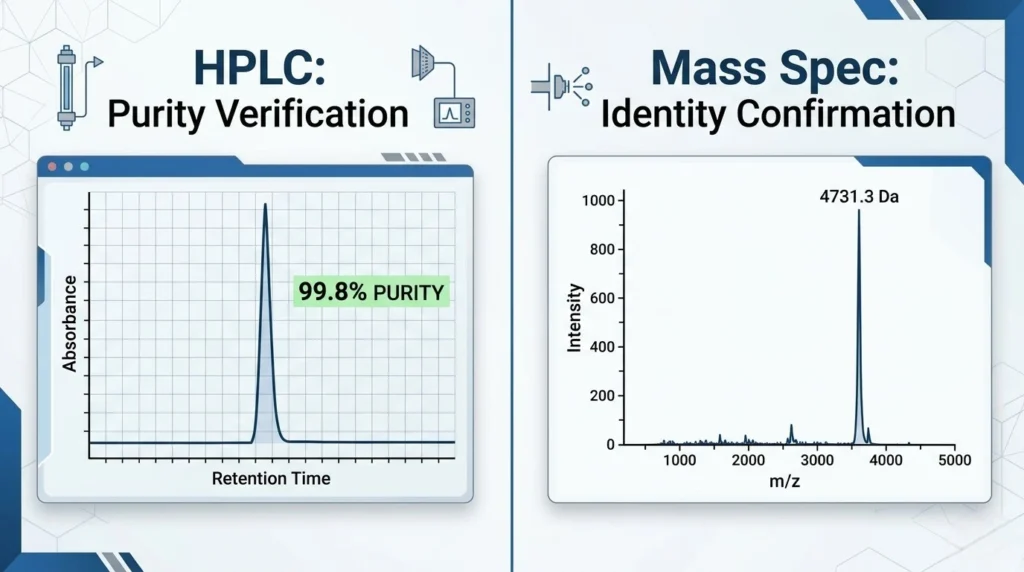
1. HPLC: Verifying Purity (The Chromatogram)
HPLC is used to determine the purity of a peptide by separating the target molecule from any impurities or residual solvents.
What to Look For:
- The Main Peak: In a high-quality sample, you should see one dominant, sharp, and symmetrical peak. This represents the target peptide.
- Peak Area Percentage: The “Purity” is calculated by the area under the main peak relative to the total area of all peaks. A report showing 99.2% means that 99.2% of the detected UV-absorbing material is your target peptide.
- Retention Time (RT): This is the time it takes for the peptide to travel through the column. While it varies by method, a consistent RT across batches indicates manufacturing stability.
2. Mass Spectrometry (MS): Verifying Identity
While HPLC tells you how pure the substance is, it doesn’t prove what the substance is. Mass Spectrometry solves this by measuring the Molecular Weight (MW) of the molecules.
How to Read the MS Report:
- The Target Mass: Every peptide has a theoretical molecular weight (e.g., Retatrutide is approximately 4731.33 Da).
- The M+H / M+Na Peaks: In an MS report, you will often see the target mass plus the weight of a hydrogen ion ($M+H$) or a sodium ion ($M+Na$). For example, if your peptide’s MW is 1000, look for a peak at 1001.0 or 1023.0.
- Interpretation: If the primary mass peak on the report matches the theoretical molecular weight of the peptide, the identity is confirmed.
3. Why 99% Purity Matters in Research
In laboratory settings, even a 2-3% impurity level can introduce “confounding variables.” These impurities are often truncated peptide sequences (shorter versions of the target) or residual TFA (Trifluoroacetic acid). These can:
- Induce Toxicity: Causing unexpected cell death in in-vitro studies.
- Alter Results: Binding to unintended receptors, leading to false-positive or false-negative data.
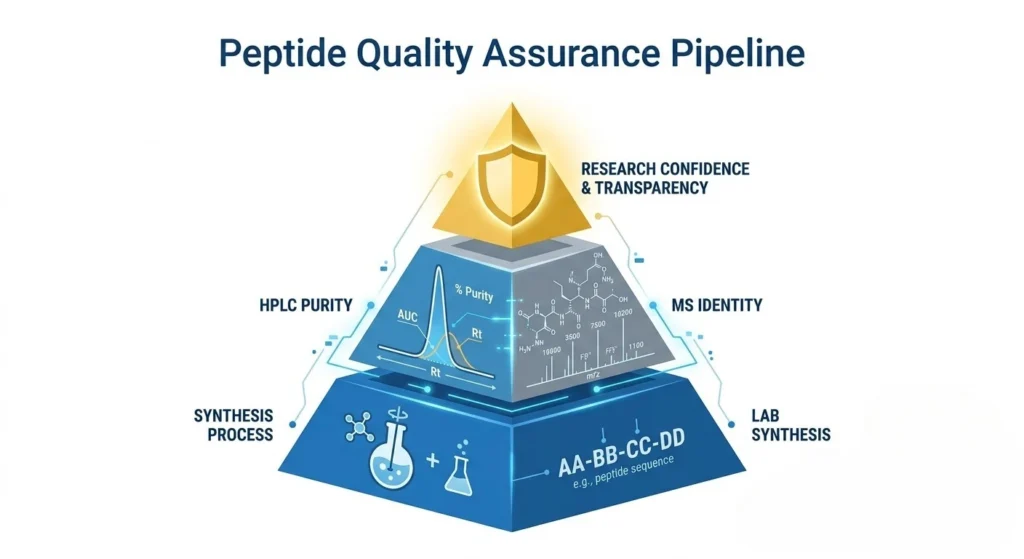
4. Our Commitment to Transparency
We believe that data should speak louder than marketing. Every batch in our catalog undergoes rigorous testing:
- Initial Synthesis Verification: Internal HPLC testing post-production.
- Third-Party Validation: Periodic independent testing to ensure cross-reference accuracy.
- Batch-Specific Tracking: Every vial is linked to a specific production run and its corresponding COA.
Research Opportunity: View our latest batch results on any product page. Our standards are consistently verified to ≥ 99% purity. Explore our for more details.
Conclusion
A Certificate of Analysis is only as good as the researcher’s ability to interpret it. By demanding and verifying HPLC and MS data, investigators ensure the integrity of their work and the safety of their research environment.
NOTE: These products are intended for laboratory research use only. This peptide is not intended for personal use. Please review and adhere to our Terms and Conditions before ordering.